Hydrogen atom have Bonding electrons = 2 (one single bond)įormal charge on iodine = (1 – 0 – 2/2) = 0 Hydrogen atom have Lone pair electrons = 00 Hydrogen atom: Hydrogen atom have Valence electrons = 01 So, the boron atom has zero formal charge in BH3 lewis structure. Let us calculate the formal charge for BH3 lewis structure.īoron atom: Boron atoms Valence electrons = 03īoron atoms Bonding electrons = 06 (three single bond)įormal charge on boron atom = (3 – 0 – 6/2) = 0 BH3 lewis structure formal chargeįormal charge calculation of any lewis structure is done by the following formula:įormal charge = (valence electrons – non-bonding electrons – ½ bonding electrons) As both H and B atoms do not contain eight electrons, so they both have incomplete octet. Thus three hydrogen atoms also has incomplete octet. Similarly all three hydrogen atoms contain two electrons (bond pair electrons). Thus, boron atom contains six electrons after bonding, two electrons present in each B-H bonds. So, all the valence electrons of BH3 lewis structure get engaged in bonding remaining no electrons for further sharing. Boron atom contains three valence electron as it is comes under 2 nd group of periodic table and hydrogen atom contains one valence electrons as it comes under the 1 st group of periodic table.īoth boron and hydrogen atoms share their valence electrons with each other to form three boron hydrogen (B-H) bonds. Octet rule tells us about the presence of complete eight electrons in its valence shell last orbital. Valence electrons in BH3 lewis structure BH3 lewis structure octet rule 
As the Born atom gains the central position due to lowest electronegativity, thus all the three hydrogen atom gets linked to boron atom. So, it has three bond pairs and zero lone electron pairs in BH3 leis structure. The BH3 lewis structure has three boron hydrogen (B-H) bonds. Then we have to make bonds between H and three H atoms to connect them with each other. Thus boron atom should be situated in the central position of the BH3 lewis structure. So, boron atom has lowest electronegativity than hydrogen atom. The Boron atom has electronegativity 2.04 and the hydrogen atom has electronegativity 2.2. In BH3 lewis structure, the boron atom belongs to 3rd group of periodic table and hydrogen atom belongs to 1st group of periodic table having 3 and 2 valence electrons respectively. To draw the lewis structure there are some rules to be followed like count total valence electrons, select the central metal atom which have lowest electronegativity, octet rule, formal charge, etc. Here, in this editorial we are learning about BH3 lewis structure. Molecular weight of Boron tri- hydride is 13.84. BH3 consists of one boron atom and three hydrogen atoms. IUPAC name for BH3 is borane which is also known as trihydrido borane. It is also known as Boranes.īH3 (borane) is comes under natural products which is originated from Erysimum inconspicuum. 
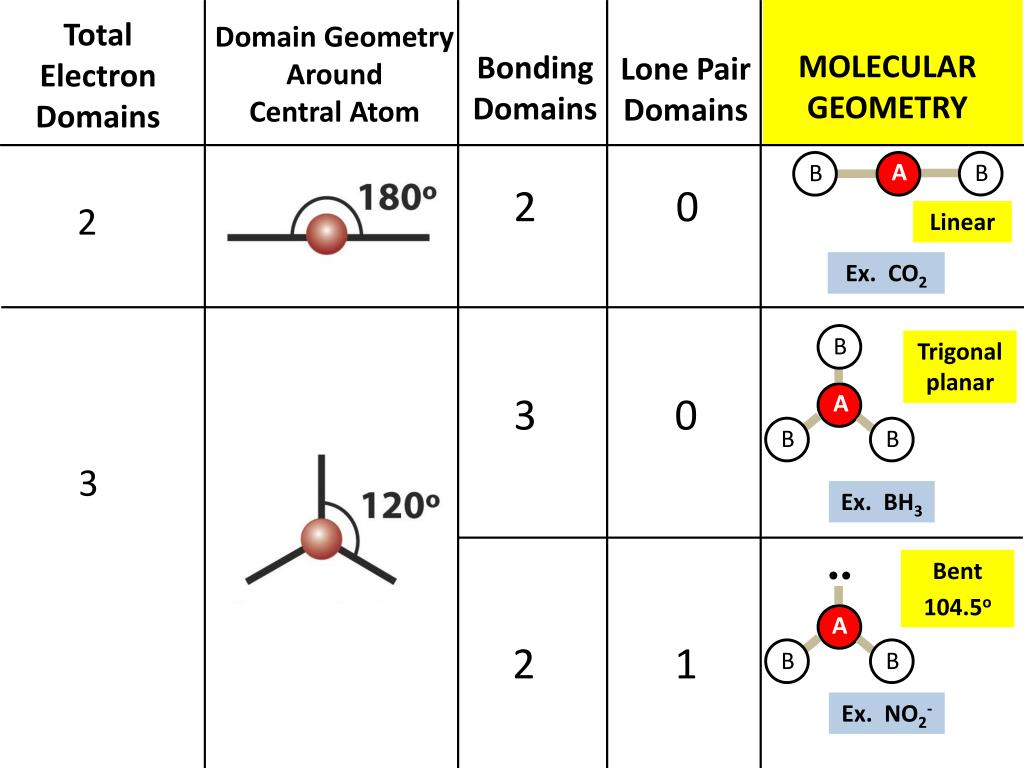
BH3 is the chemical formula of Boron tri- hydride.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
